Michelet 2013 Front Plant Sci
| Michelet L, Zaffagnini M, Morisse S, Sparla F, Pérez-Pérez ME, Francia F, Danon A, Marchand CH, Fermani S, Trost P, Lemaire SD (2013) Redox regulation of the Calvin-Benson cycle: something old, something new. Front Plant Sci 4:470. https://doi.org/10.3389/fpls.2013.00470 |
Michelet L, Zaffagnini M, Morisse S, Sparla F, Pérez-Pérez ME, Francia F, Danon A, Marchand CH, Fermani S, Trost P, Lemaire SD (2013) Front Plant Sci
Abstract: Reversible redox post-translational modifications such as oxido-reduction of disulfide bonds, S-nitrosylation, and S-glutathionylation, play a prominent role in the regulation of cell metabolism and signaling in all organisms. These modifications are mainly controlled by members of the thioredoxin and glutaredoxin families. Early studies in photosynthetic organisms have identified the Calvin-Benson cycle, the photosynthetic pathway responsible for carbon assimilation, as a redox regulated process. Indeed, 4 out of 11 enzymes of the cycle were shown to have a low activity in the dark and to be activated in the light through thioredoxin-dependent reduction of regulatory disulfide bonds. The underlying molecular mechanisms were extensively studied at the biochemical and structural level. Unexpectedly, recent biochemical and proteomic studies have suggested that all enzymes of the cycle and several associated regulatory proteins may undergo redox regulation through multiple redox post-translational modifications including glutathionylation and nitrosylation. The aim of this review is to detail the well-established mechanisms of redox regulation of Calvin-Benson cycle enzymes as well as the most recent reports indicating that this pathway is tightly controlled by multiple interconnected redox post-translational modifications. This redox control is likely allowing fine tuning of the Calvin-Benson cycle required for adaptation to varying environmental conditions, especially during responses to biotic and abiotic stresses.
• Bioblast editor: Gnaiger E
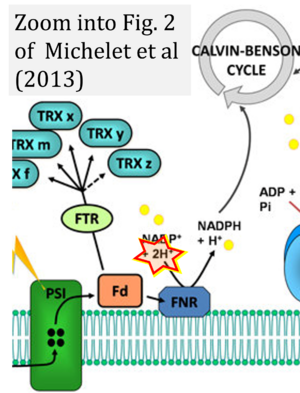
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.